|
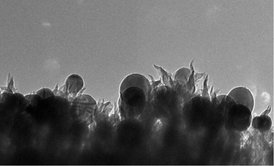
In a new twist to waste-to-fuel technology, scientists at the Department of Energy's Oak Ridge National Laboratory have developed an electrochemical process that uses tiny spikes of carbon and copper to turn carbon dioxide, a greenhouse gas, into ethanol. Their finding, which involves nanofabrication and catalysis science, was serendipitous. "A process like this would allow you to consume extra electricity when it's available to make and store as ethanol," Rondinone said. "This could help to balance a grid supplied by intermittent renewable sources." Given the technique's reliance on low-cost materials and an ability to operate at room temperature in water, the researchers believe the approach could be scaled up for industrially relevant applications. For instance, the process could be used to store excess electricity generated from variable power sources such as wind and solar.
0 Comments
Leave a Reply. |
Archives
November 2016
Categories |
 RSS Feed
RSS Feed
